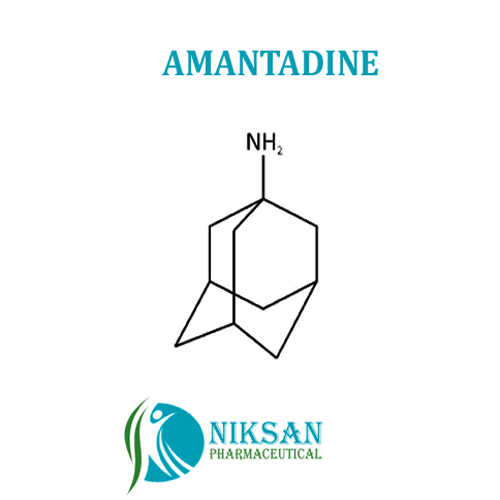
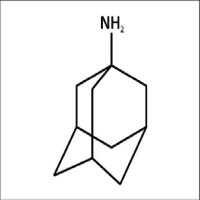
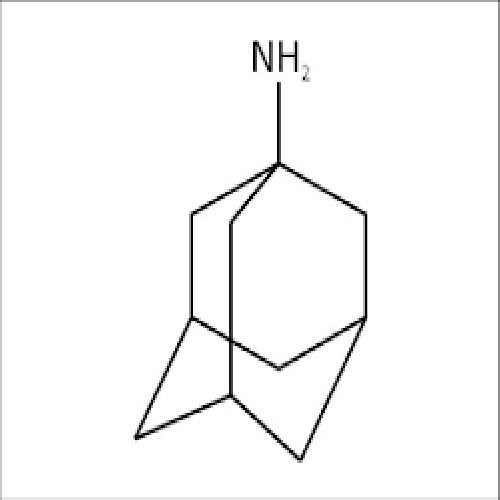
AMANTADINE
Product Details:
- Boiling point not explicitly
- Melting Point greater than 300C
- Molecular Formula C10H17NHCl
- HS Code 30049099
- Storage Room Temperature
- Shelf Life 3 Years
- EINECS No 212-201-2
- Click to View more
AMANTADINE Price And Quantity
- 6500.00 INR/Kilograms
- 1 Kilograms
AMANTADINE Product Specifications
- white or nearly white, crystalline powder
- not explicitly
- Pharmaceutical Intermediates
- greater than 300C
- C10H17NHCl
- 30049099
- Room Temperature
- 3 Years
- tricyclo[3.3.1.13,7]decan-1-amine
- 212-201-2
- AMANTADINE
- Other
- not contain
- Medicine Grade
- 1.2-micrometer mass median diameter
- C10H17NHCl
- 98%
- Powder
- not more than 0.5%
- 151.25 g/mol Grams (g)
- 665-66-7
- between 4.0 and 6.0
- freely soluble in water and in ethanol
- Bitter
AMANTADINE Trade Information
- Mumbai
- 100 Kilograms Per Week
- 5 Days
- No
- HDPE DRUM WITH TWO LDPE INNER LINER
- Dadra and Nagar Haveli, Chandigarh, Himachal Pradesh, Andaman and Nicobar Islands, Uttarakhand, Daman and Diu, Lakshadweep, Nagaland, South India, North India, East India, West India, Andhra Pradesh, Assam, Arunachal Pradesh, Bihar, Delhi, Goa, Jammu and Kashmir, Jharkhand, Karnataka, Maharashtra, Mizoram, Meghalaya, Manipur, Pondicherry, Rajasthan, Sikkim, Tamil Nadu, Telangana, Tripura, West Bengal, Haryana, Gujarat, Uttar Pradesh, Madhya Pradesh, Punjab, Kerala, Central India, Odisha, Chhattisgarh, All India
- WHO GMP,GMP,GLP,ISO
Product Description
is one of the biggest manufacturer, supplier, exporter and trader ofthe AMANTADINEfinished formulations moreover AMANTADINE API. Niksan Pharmaceutical is the huge manufacturer, exporter andsupplier of AMANTADINE API and formulation situated in Ankleshwar, Gujarat, India.
NiksanPharmaceutical is the suppliers, manufacturer, exporters and trader of
AMANTADINE inthedomestic level as well as the international market.
Niksan is anIndian based pharmaceutical company so it also supplies and manufactures the AMANTADINE to allIndian states like Gujarat, Maharashtra, Punjab, Delhi, Tamilnadu, Goa, UttarPradesh, Karnataka, Jammu & Kashmir, West Bengal, Assam, Rajasthan,Hyderabad, Karnataka, Kerala, Madhya Pradesh, Manipur,Meghalaya, Mizoram, Nagaland, Orissa, Punjab and Haryana.
NiksanPharmaceutical and Niksan group companies are exporters, suppliers andmanufacturers of the API and also AMANTADINE formulations in many countries for many years. Niksanpharmaceutical currently exports AMANTADINE API and Formulation to countries like New Zealand, Jordan,Belgium, United Kingdom, Australia, Italy, Ireland, Hong Kong, Morocco,,Portugal, France, South Africa, Philippines, Malaysia, Singapore, Israel,Greece, Saudi Arabia, Egypt, Taiwan, Netherlands, South Korea, Switzerland,Algeria, Pakistan, Canada, Vietnam, United States, Germany, Brazil and manyother countries.
AMANTADINEis an antiviral andantiparkinsonian medication. Initially developed to prevent and treat influenzaA, it is now also used in the management of Parkinsons disease anddrug-induced extrapyramidal symptoms.
SYNONYSMS:1-Adamantanamine, Symmetrel
IUPAC NAME: 1-Adamantanamine
CAS NO:768-94-5
FORMULA:C10H17N
MOLECULAR MASS: 151.25 g/mol
STORAGE CONDITIONS:Store at room temperature between 20C to25C (68F to 77F). Protect from moisture and light. Keep container tightlyclosed.
HOW TO USE:
For Influenza A prophylaxis/treatment:100 mg orally twice daily for adults; 100 mg daily for elderly or renalimpairment.
FOR Parkinsons disease: Initial dose is usually 100 mg once daily,increasing to 100 mg twice daily after 1 week.
Follow your doctors prescription carefully.
HOW AMANTADINEWORKS:Amantadineworks by blocking the M2 proton channel of the influenza A virus, preventingviral uncoating inside host cells.
In Parkinsons disease, it enhances dopamine release, blocks dopamine reuptake,and exerts mild anticholinergic effects, improving motor symptoms.
PHARMACOKINETICSOF AMANTADINE: AMANTADINE Well absorbed orally. idely distributed, crosses the blood-brainbarrier. Minimal hepatic metabolism. Primarily excreted unchanged by thekidneys. 1015 hours (may be longer in elderly or renal impairment).
SIDE EFFECTSOF AMANTADINE:
Common: Nausea, dizziness, insomnia, dry mouth,constipation.
Serious: Hallucinations, confusion, orthostatic hypotension, livedoreticularis, seizures.
PRECAUTIONS: Avoid suddendiscontinuation in Parkinsons patients (may worsen symptoms). Use cautiouslyin elderly, renal impairment, seizure disorders, and psychiatric conditions.May cause drowsinessavoid driving if affected.
CDSCOAPPROVAL:
Amantadine Hydrochloride 1972-December
FORMULATIONSAVAILABLE IN MARKET:
Amantadine 100 mg Capsules
Amantadine Hydrochloride Oral Solution
Note: Productprotected by valid patents are not offered for sale in countries where suchpatents are still valid and its liability is at Buyers Risk
REFERENCES:
www.webmd.com
https://pubchem.ncbi.nlm.nih.gov
https://go.drugbank.com
https://cdscoonline.gov.in
https://www.wikipedia.org/
https://www.drugs.com
https://www.zaubacorp.com
https://www.practo.com/consult
Exceptional Pharmaceutical Quality
AMANTADINE is produced under strict quality controls, ensuring a purity of not less than 98%. The raw material is supplied as a fine, crystalline powder and serves as a vital intermediate in the production of essential medicines. Its bitter taste and high solubility make it versatile for various pharmaceutical applications.
Optimal Storage and Shelf Life
To preserve stability and effectiveness, AMANTADINE should be stored in a cool, dry environment away from direct sunlight and moisture. The product maintains its integrity for up to three years, making it suitable for long-term pharmaceutical use if kept under recommended conditions.
FAQs of AMANTADINE:
Q: How should AMANTADINE be stored to maintain its quality?
A: It is important to keep AMANTADINE in a cool, dry place, away from moisture and direct light, ideally at room temperature to preserve its potency and stability.Q: What are the primary uses of AMANTADINE in pharmaceuticals?
A: AMANTADINE is primarily used as a pharmaceutical intermediate in the synthesis of various medicines, due to its high purity and solubility in water and ethanol.Q: When does AMANTADINE expire, and how long does its shelf life last?
A: When stored correctly, AMANTADINE has a shelf life of up to 3 years from the date of manufacture, as long as it remains in its original, sealed packaging.Q: Where can AMANTADINE be sourced from?
A: AMANTADINE is available through reputable distributors, exporters, manufacturers, and suppliers in the pharmaceutical sector, particularly in India.Q: What process is involved in using AMANTADINE as a medicine intermediate?
A: AMANTADINE is integrated during pharmaceutical production for the synthesis of active drug compounds, selected for its chemical stability and precise particle size.Q: How can the solubility of AMANTADINE benefit pharmaceutical formulations?
A: AMANTADINEs high solubility in water and ethanol enhances its compatibility and effectiveness in creating homogeneous formulations for various medical applications.
Price:
- 50
- 100
- 200
- 250
- 500
- 1000+
Other Products in 'Active Pharmaceuticals Ingredients (API)' category
 |
NIKSAN PHARMACEUTICAL
All Rights Reserved.(Terms of Use) Developed and Managed by Infocom Network Private Limited. |
 English
English Spanish
Spanish French
French German
German Italian
Italian Chinese (Simplified)
Chinese (Simplified) Japanese
Japanese Korean
Korean Arabic
Arabic Portuguese
Portuguese