FE NOFIBRATE
Product Details:
- Taste Bitter
- Boiling point 469.8 C
- Ph Level pH 1.2, 6.8, and 7.4
- Smell Other
- Molecular Formula C20H21ClO4
- Particle Size 169.6 m
- Heavy Metal (%) 0.1%
- Click to View more
FE NOFIBRATE Price And Quantity
- 5 Kilograms
FE NOFIBRATE Product Specifications
- Medicine Grade
- 3 Years
- 29420090
- 99 %
- Keep away from moisture
- 360.83 g/mol Grams (g)
- White to Off-White Solid
- 256-376-3
- practically insoluble in water
- C20H21ClO4
- 0.1%
- 79-82C
- not exceeding 0.5%
- 169.6 m
- FENOFIBRATE
- Other
- Powder
- pH 1.2, 6.8, and 7.4
- C20H21ClO4
- 49562-28-9
- Pharmaceutical Intermediates
- Bitter
- 469.8 C
- FENOFIBRATE
- Fenofibrate comes in the anti lipidemic class of medication. Fenofibrate lowers the LDL (low density lipoproteins) which is also called bad cholesterol. Fenofibrate increase the natural substance which cause degradation of the fats in the blood.
FE NOFIBRATE Trade Information
- INDIA
- Cash Advance (CA), Cheque
- 100 Kilograms Per Month
- 1 Days
- Yes
- Free samples are available
- HDPE DRUM WITH TWO LDPE INNER LINER
- Dadra and Nagar Haveli, Chandigarh, Himachal Pradesh, Andaman and Nicobar Islands, Uttarakhand, Daman and Diu, Lakshadweep, Chhattisgarh, South India, Nagaland, East India, Andhra Pradesh, Assam, Arunachal Pradesh, Bihar, Goa, Haryana, Jammu and Kashmir, Jharkhand, Karnataka, Madhya Pradesh, Mizoram, Meghalaya, Manipur, Punjab, Pondicherry, Rajasthan, Sikkim, Tamil Nadu, Telangana, Tripura, West Bengal, Maharashtra, Delhi, Gujarat, Uttar Pradesh, North India, Kerala, Central India, Odisha, West India, All India
- WHO GMP,GMP,GLP,ISO
Product Description
NiksanPharmaceutical and our group companies Niksan healthcare are the one of largestmanufacturer, exporter and supplier of Fenofibrateformulations and Fenofibrate API in Ankleshwar, Gujarat, India. We aresullying the best quality of Fenofibrate API all around the India as well as inthe whole world. Our product Fenofibrate praised by our clients and also by theother companies.
Niksan Pharmaceutical is theworld leading manufacturing and supplying company of the Fenofibrate products and API.
Thus we are Indian based company we supplying our best quality products ofFenofibrate to every states of India formany years like Jammu and Kashmir, Kerala, Punjab, Rajasthan, Andhra Pradesh,Haryana, Telangana, Bihar, Karnataka, Delhi, Tamil Nadu, Odisha, Maharashtra,West Bengal, Uttar Pradesh, Gujarat, Madhya Pradesh, Himachal Pradesh, Goa,Chhattisgarh etc.
NiksanPharmaceutical also export huge quantity of best quality Fenofibrate API products tothe other nations like Puerto Rico,Philippines, United State, Nepal, Singapore, Lebanon, Indonesia, India,Bangladesh, Canada, United Arab Emirates, Sri Lanka, France, Jordan, Taiwan ,Malaysia, Vietnam, Thailand, Saudi Arabia, Australia , Pakistan, Iraq, SouthKorea, Hong Kong, Ireland, United Kingdom Algeria, Egypt, Belgium, Iran,Israel, Mexico, Germany, Turkey, Japan, Brazil and many south easterncountries.
Fenofibrate comes in the anti lipidemic class of medication. Fenofibrate lowers theLDL (low density lipoproteins) which is also called bad cholesterol. Fenofibrate increase the natural substancewhich cause degradation of the fats in the blood.
SYNONYMS: Fenofibrat, Fenofibrato, Fenofibratum,Finofibrate, Procetofen.
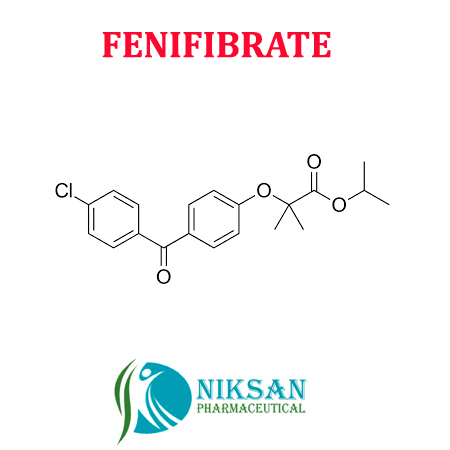
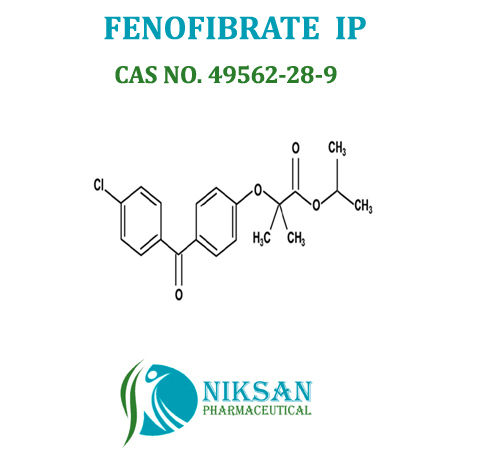
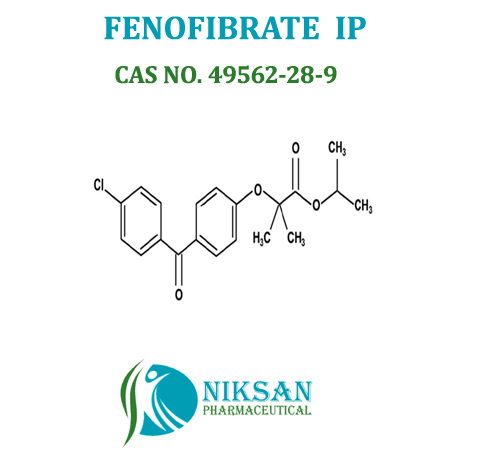
IUPAC NAME: Propan-2-yl 2-[4-(4-chlorobenzoyl) phenoxy]-2-methylpropanoate
CAS NO: 49562-28-9
FORMULA:C20H21ClO4
MOLECULAR MASS: 360.83 g/mol
STORAGE CONDITIONS: Store in cool and dry place, keep away from directheat and light. Do not store in bathroom or kitchen. Keep away from children and pet.
HOW TO USE: Take Fenofibrate by mouth one time per daywith or without food. Take medicine with food or milk if you feel stomachupset. Take your doctors advice if you have any confusion about medicine.
HOW FENOFIBRATEWORKS: Fenofibrate lowers the bad cholesterol levelby increasing the development of natural enzymes who breaks the fat or LDL inthe blood. Fenofibrate lowers the bad cholesterols like LDL and triglycerideand increase the good cholesterol like HDL in body.
PHARMACOKINETICS: Fenofibrate rapidly absorbed in the bodyafter the oral administration. It takes Fenofibrate 4-6 hours to reach the peakplasma concentration. Almost 99% of Fenofibrate binds with the blood plasmaproteins. The half-life of Fenofibrate is about 19-27 hours in the normalpatient and 143 hours in the patient with renal failure. The 60-88%of Fenofibrateeliminate through urination and 5-25% if Fenofibrate eliminate by the feces.
SIDE EFFECTS OF FENOFIBRATE: There are no many side effects of Fenofibrateis seen in the patient. Some side effects like kidney stone and liver problemsare seen in some patients. Tell your doctor if you see some side effects likenausea and vomiting, stomach or intestinal pain, abdominal irritation, yelloweyes or skin and black urine. This drug rarely because effects like musclepain, weakness in body and kidney problems.
PRECAUTIONS: The main precaution is keep check your HDLlevel. Tell your doctor if you have allergic reactions toward the Fenofibrate medicationor its metabolite. If you have and disease like kidney problem, abdominalproblem, liver problem or heart problem tell your doctor before writing theprescription. Do not take medication if you are in pregnancy period or inlactation period.
CDSCO APPROVAL: Rosuvastatin Calcium eq. to Rosuvastatin20mg/20mg + Fenofibrate BP 67.5mg/145mg Tablets approved by CDSCO in Indian in24.08.2011,
Atorvastatin + Fenofibrate approved by CDSCO in Indian in24.12.2004,
Atorvastatin 5mg + Fenofibrate 160mg (additional strength)approved by CDSCO in Indian in 01.12.2006,
Atorvastatin Calcium (10mg) + Fenofibrate (145mg) (addl. strength)approved by CDSCO in Indian in 30.10.2007,
Ezetimibe 10 mg + Fenofibrate 145 mg Film coated tablets approvedby CDSCO in Indian in 01.09.2009,
FDC of Atorvastatin + fenofibrate approved by CDSCO in Indian in01.08.2006,
Rosuvastatin 5/10/20mg + Fenofibrate 67/145/160mg approved byCDSCO in Indian in 23.08.2010
Choline Fenofibrate DR Capsule 45mg/135mg & DR Tablet45mg/135mg. approved by CDSCO in Indian in 01.11.2012,
Rosuvastatin Calcium IP 5mg/10mg + Fenofibrate BP 160mg/160mgtablet approved by CDSCO in Indian in 29.12.2010,
Atorvastatin 10mg + Fenofibrate 80mg tablet (AdditionalIndication) 25.01.2008,
Metformin HCl ER 500mg + Fenofibrate 80mg/160mg tablet approved byCDSCO in Indian in 25.01.2008,
Fenofibrate capsules approved by CDSCO in Indian in 22.12.1999,
Ezetimibe 10mg + Fenofibrate 160mg tablet approved by CDSCO inIndian in 04.04.2007
FORMULATIONS AVAILABLE IN MARKET:
Fenofibrate 45mg tablets
Fenofibrate 35mg tablets
Fenofibrate 67mg tablets
Ezetimibe 10mg + Fenofibrate 160mg tablets
Fenofibrate capsules
Atorvastatin 10mg + Fenofibrate 80mg tablets
Rosuvastatin Calcium IP 5mg + Fenofibrate BP 160mg tablets
Rosuvastatin Calcium IP10mg + Fenofibrate BP 160mg tablets
Ezetimibe 10 mg + Fenofibrate 145 mg Film coated tablets
Atorvastatin 5mg + Fenofibrate 160mg tablets
Note: Product protected by valid patents are notoffered for sale in countries where such patents are still valid and itsliability is at Buyers Risk
REFERENCES:
www.webmd.com
https://pubchem.ncbi.nlm.nih.gov
https://go.drugbank.com
https://cdscoonline.gov.in
https://www.wikipedia.org/
https://www.drugs.com
https://www.zaubacorp.com
https://www.practo.com/consult
Powerful Cholesterol Management
Fenofibrate is classified in the anti-lipidemic medication group, specifically designed to reduce harmful LDL cholesterol and triglycerides. It works by activating natural enzymes that help degrade fats in the blood, contributing to improved lipid profiles and lowering the risk of cardiovascular problems.
High Purity and Quality Control
Manufactured to strict pharmaceutical standards, Fenofibrate powder features a purity of 99% and contains no more than 0.5% loss on drying or 0.1% heavy metals. Its white to off-white appearance indicates quality, and with a shelf life of three years, it assures long-term reliability for formulators.
Easy Storage and Handling
To maintain its efficacy, Fenofibrate should be kept in a cool, dry place, away from moisture, direct light, and heat. Storing it outside of kitchens and bathrooms ensures product stability and safety, facilitating its use across different manufacturing environments.
FAQs of FENOFIBRATE:
Q: How should Fenofibrate powder be stored to ensure its longevity?
A: Fenofibrate powder should be stored in a cool and dry area, away from direct heat and light, and outside of kitchens or bathrooms to prevent exposure to moisture, which can compromise its stability and effectiveness.Q: What is the process for using Fenofibrate in medicinal formulations?
A: Fenofibrate is typically incorporated into tablets or capsules by pharmaceutical manufacturers. Its powder form allows for precise dosing and easy mixing with other excipients during the formulation process.Q: When is the best time to take Fenofibrate for optimal results?
A: Fenofibrate should be taken once daily by mouth, with or without food. If stomach upset occurs, taking it with food or milk is recommended to improve tolerability.Q: Where is Fenofibrate most commonly utilized as a raw material?
A: Fenofibrate is widely used in pharmaceutical manufacturing, particularly by distributors, exporters, importers, traders, and suppliers in India for the production of anti-lipidemic tablets and capsules.Q: What are the main benefits of Fenofibrate for cardiovascular health?
A: Fenofibrate effectively lowers bad cholesterol (LDL) and triglyceride levels, while boosting the activity of natural substances in the body that break down fats. This can help reduce the risk of heart disease and related complications.Q: How does Fenofibrate simplify digestion and lipid breakdown?
A: Fenofibrate enhances the development of enzymes that facilitate the degradation of fats within the bloodstream, supporting efficient lipid metabolism and improving overall digestive processes.
Price:
- 50
- 100
- 200
- 250
- 500
- 1000+
Other Products in 'Active Pharmaceuticals Ingredients (API)' category
 |
NIKSAN PHARMACEUTICAL
All Rights Reserved.(Terms of Use) Developed and Managed by Infocom Network Private Limited. |
 English
English Spanish
Spanish French
French German
German Italian
Italian Chinese (Simplified)
Chinese (Simplified) Japanese
Japanese Korean
Korean Arabic
Arabic Portuguese
Portuguese