CILNIDIPINE
Product Details:
- Ph Level 7
- Boiling point 652.6
- Melting Point 110
- Shelf Life 5 Years
- Molecular Formula C27H28N2O7
- EINECS No 132203-70-4
- HS Code 29420090
- Click to View more
CILNIDIPINE Price And Quantity
- 6000.0 INR/Kilograms
- 5 Kilograms
CILNIDIPINE Product Specifications
- 99.8 %
- SOLUBLE IN PHARMACEUTICAL SOLVENT
- CILNIDIPINE
- 132203-70-4
- Odorless
- CILNIDIPINE
- Room Temperature
- 0.5
- Cilnidipine comes under the calcium channel blocker class of medicine. Most of it is used as anti-hypertensive agent. Cilnidipine have compatibility with other API like Telmisartan, Olmesartan Medoxomil, Ramipril and Metoprolol Succinate. Cilnidipine can act on the N-type calcium channel in addition to acting on the L-type calcium channel.Cilnidipine dilates both arterioles and venules, reducing the pressure in the capillary bed.
- 110
- YELLOW POWDER
- 5 Years
- 652.6
- 7
- C27H28N2O7
- Cardiovascular Agents
- 492.52 Grams (g)
- 20 MICRON
- Woody
- 10 PPM
- Medicine Grade
- 132203-70-4
- C27H28N2O7
- Powder
- 29420090
CILNIDIPINE Trade Information
- INDIA
- 100 Kilograms Per Month
- 1 Days
- No
- Within a certain price range free samples are available
- HDPE DRUM WITH TWO LDPE INNER LINER
- Asia, Middle East, Africa
- All India, South India, Central India, West India, North India, East India, Gujarat, Karnataka, Kerala, Lakshadweep, Mizoram, Goa, Jharkhand, Odisha, Punjab, Assam, Delhi, Dadra and Nagar Haveli, Andaman and Nicobar Islands, Arunachal Pradesh, Chhattisgarh, Haryana, Himachal Pradesh, Jammu and Kashmir, Madhya Pradesh, Maharashtra, Nagaland, Rajasthan, Sikkim, Tamil Nadu, Telangana, Tripura, Pondicherry, Uttar Pradesh, Uttarakhand, West Bengal, Meghalaya, Manipur, Andhra Pradesh, Bihar, Chandigarh, Daman and Diu
- FDCA, GMP, GLP AND ISO
Product Description
NIKSAN PHARMACEUTICAL providesCilnidipineto all states of India like Punjab, Rajasthan, Andhra Pradesh, Haryana, Telangana, Bihar, Karnataka, Delhi, Tamil Nadu, Odisha, Maharashtra, West Bengal, Uttar Pradesh, Gujarat, and Madhya Pradesh etc. We also give gift samples to the companies for R&D purpose.Cilnidipinecomes under the calcium channel blocker class of medicine. Most of it is used a santi-hypertensive agent.
Cilnidipine have compatibility with other API like Telmisartan, Olmesartan Medoxomil, Ramipril and Metoprolol Succinate. Cilnidipine can act on theN-type calcium channel in addition to acting on theL-type calcium channel. Cilnidipinedilates both arterioles and venues, reducing the pressure in the capillary bed.
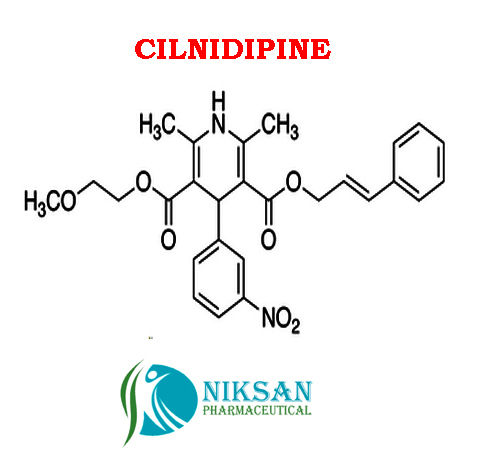
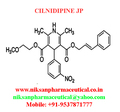
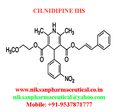
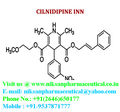
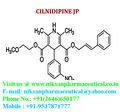
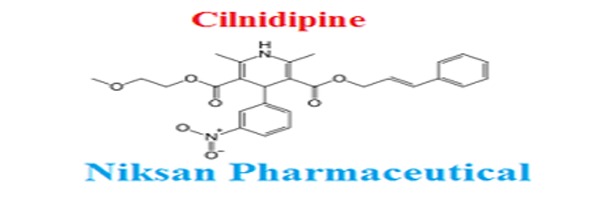
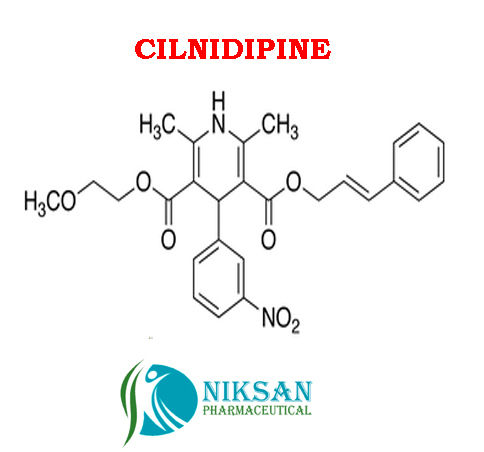
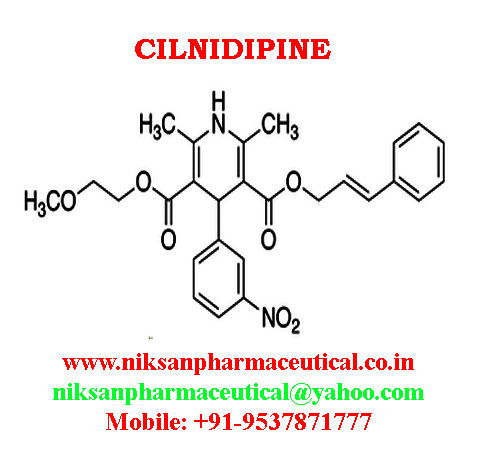
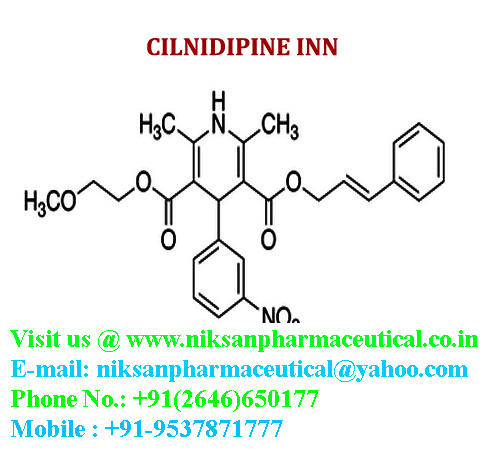
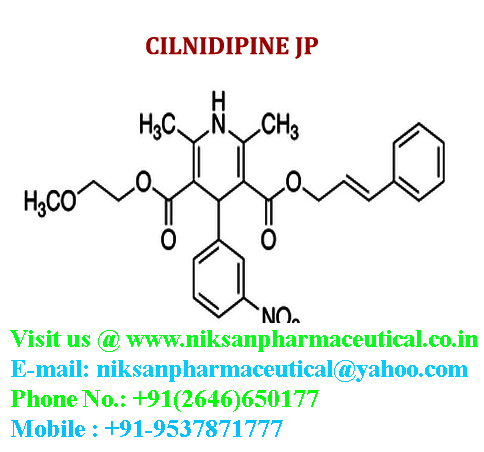
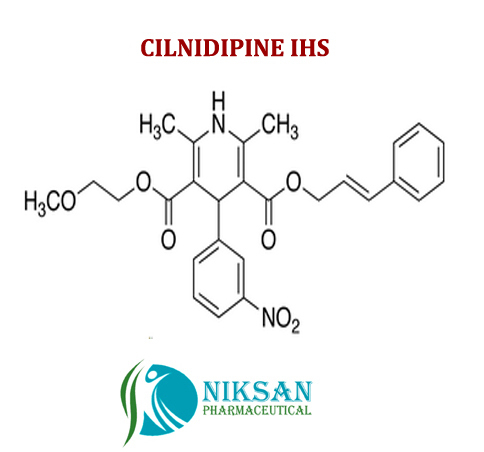
IUPAC NAME:3-(2-methoxyethyl)5-(2E)-3-phenylprop-2-en-1-yl2,6-dimethyl-4-(3-nitrophenyl)-1,4-dihydropyridine-3,5-dicarboxylate
CASNO:132203-70-4
FORMULA:C27H28N2O7
MOLECULAR MASS:492.52 g/mol
STORAGE:Store the medicine in cool and dry place. Store it away from the direct sun light. Do not change packet of the Cilnidipine tablet. Keep away from reach of children and pets.
APPLICATION:Cilnidipine is primarily used for the treatment of high blood pressure also referred to as hyper tension. The Cilnidipine have the calcium channel blocker activity it is also used in the treatment of headache, vision related problems, irregular heartbeat, chest pain, and fatigue.
HOW TO USE:Simply take the medicine as per your doctor says. The tablet should be taken orally with water. The tablet must be swallowed whole without breaking or chewing.
HOW CILNIDIPINE WORKS:Cilnidipine blocks the calcium channels in blood vessels. By this the blood supplies towards the heart increase and by that the hyper tension decreases.
CONTRAINDICATIONS:Do not take Cilnidipine medicine in pregnancy and lactation period. Do not give medicine to the patient under 18 years. If you have any confusion related to the medicine consult to your doctor.
PHARMACOKINETICS: Cilnidipine is one type of lipophilic drug. So mainly itis metabolites in the liver or kidneys. Cilnidipine is rapidly absorbed in the blood plasma in only 2hrs . Cilnidipine normally excreted by urination.
SIDE EFFECTS:There are some common side effects of the Cilnidipine are Dizziness, hypotension, swelling of (face, eyes, tongue, and feet), fast heartbeats, muscle pain, stomach pain, skin rash.
PRECAUTIONS:Dont take medicine if you are allergic to Cilnidipine. If you have any heart or liver problem kindly find some alternative of Cilnidipine. The effect of Cilnidipine is not known yet so do not take it if you are breast feeding the baby. Do not share the medicine with other people.
CDSCO APPROVAL: Cilnidipine tablet 5mg approved by CDSCO in India in21.06.2007.
Cilnidipine tablet 10 mg is approved by CDSCO in 21.06.2007.
FORMULATIONSIN MARKET:
Cilnidipine 5 mg Tablets
Cilnidipine 10 mg Tablets
Cilnidipine 20 mg Tablets
Cilnidipine 5 mg +Telmisartan 20 mg Tablets
Cilnidipine10 mg +Telmisartan 40 mg Tablets
Cilnidipine 10 mg +Metoprolol Succinate 25mg Tablets
Cilnidipine 10 mg +Metoprolol Succinate 50mg Tablets
Cilnidipine 10 mg +Olmesartan Medoxomil 20mg Tablets
Cilnidipine 10 mg +Olmesartan Medoxomil 40mg Tablets
Cilnidipine 10 mg +Ramipril IP 10 mg Capsules
Cilnidipine 10 mg +Ramipril IP 2.5 mgCapsules
Cilnidipine 5 mg +Hydrochlorothiazide 12.5mg + Telmisartan 40 mg capsules
Note:Product protected by valid patents are not offered for sale in countries where such patents are still valid and its liability is at Buyers Risk
How to Take Cilnidipine Tablets or Capsules
Cilnidipine tablets or capsules should be taken exactly as prescribed by your healthcare provider. Swallow the tablet whole with water, without breaking, chewing, or crushing it. Follow the exact dosage and timing instructions from your doctor to ensure the best therapeutic outcome and to minimize possible side effects.
Proper Storage for Maximum Effectiveness
To maintain Cilnidipines potency, store the tablets or capsules in a well-closed container. Place it in a cool, dry environment and avoid exposure to direct sunlight. Do not transfer the medicine to another packet, as this may compromise its effectiveness and safety. Always follow the storage recommendations provided with your medication.
Primary Uses and Benefits
Cilnidipine is predominantly used as an anti-hypertensive agent, controlling blood pressure and reducing strain on the heart and blood vessels. Its also helpful for treating headaches, vision issues, irregular heartbeat, chest pain, and fatigue related to hypertension. By promoting blood supply and relaxing blood vessels, Cilnidipine helps improve overall cardiovascular health.
FAQs of CILNIDIPINE:
Q: How should I take Cilnidipine tablets or capsules?
A: Take Cilnidipine exactly as directed by your doctor. Swallow the tablet or capsule whole with water, preferably at the same time each day. Do not chew, break, or crush the medicine.Q: What are the recommended dosages of Cilnidipine?
A: Cilnidipine is available in 5 mg, 10 mg, and 20 mg strengths. Your doctor will determine the most suitable dosage for you based on your health condition and response to treatment.Q: When is Cilnidipine prescribed?
A: Cilnidipine is commonly prescribed to manage high blood pressure, but it may also be used for conditions such as headaches, vision problems, irregular heartbeat, chest pain, and fatigue associated with hypertension.Q: Where should I store Cilnidipine tablets or capsules?
A: Keep Cilnidipine in a well-closed container at room temperature, in a cool and dry place. Store it away from sunlight and moisture, and avoid changing the original packaging.Q: What is the process if I miss a dose of Cilnidipine?
A: If you miss a dose, take it as soon as you remember. If its almost time for your next dose, skip the missed dose and continue with your regular schedule. Avoid doubling up on doses.Q: What are the benefits of using Cilnidipine for blood pressure management?
A: Cilnidipine helps to lower blood pressure by relaxing and dilating blood vessels, promoting better blood flow, and reducing the workload on the heart. This decreases the risk of heart-related complications.
Price:
- 50
- 100
- 200
- 250
- 500
- 1000+
Other Products in 'Active Pharmaceuticals Ingredients (API)' category
 |
NIKSAN PHARMACEUTICAL
All Rights Reserved.(Terms of Use) Developed and Managed by Infocom Network Private Limited. |
 English
English Spanish
Spanish French
French German
German Italian
Italian Chinese (Simplified)
Chinese (Simplified) Japanese
Japanese Korean
Korean Arabic
Arabic Portuguese
Portuguese