LE VOSULPIRIDE
Product Details:
- Molecular Weight 341.4 g/mol Grams (g)
- Taste Other
- Particle Size 100-200 nm
- Boiling point not available
- Smell Other
- Solubility freely soluble in Methanol, 0.1 N NaOH and 0.1 N HCl
- Loss on Drying not exceeding 0.5%
- Click to View more
LE VOSULPIRIDE Price And Quantity
- 6000 INR/Kilograms
- 50 Kilograms
LE VOSULPIRIDE Product Specifications
- 3 Years
- 233-599-4
- White to Off-White Solid
- 23672-07-3
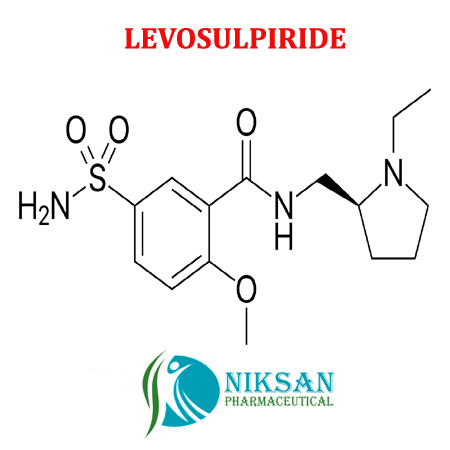
- LEVOSULPIRIDE
- C15H23N3O4S
- no more than 10 ppm
- 29420090
- Powder
- C15H23N3O4S
- between 184C and 189C
- 100-200 nm
- 341.4 g/mol Grams (g)
- Other
- Medicine Grade
- 99 %
- 3.3
- Room Temperature
- not exceeding 0.5%
- freely soluble in Methanol, 0.1 N NaOH and 0.1 N HCl
- LEVOSULPIRIDE
- Other
- Other
- not available
- Levosulpiride is antipsychotic agent which is used in the mood elevating treatments like mood swings, depression. Levosulpiride also used in the treatment to improve the food movements and gut movements.
LE VOSULPIRIDE Trade Information
- SAHAR AIR CARGO
- Cash Advance (CA), Cheque
- 100 Kilograms Per Day
- 1 Days
- No
- HDPE DRUM WITH TWO INNER LDPE LINNER
- Asia, Australia, Central America, North America, South America, Eastern Europe, Western Europe, Middle East, Africa
- All India
- FDCA, GMP, GLP AND ISO
Product Description
NiksanPharmaceutical is the largest manufacturer, Exporter and supplier of the levosulpiride. NowadaysNiksan Pharmaceutical is one of the leading manufacturer and exporter whomanufacturing levosulpiride as well as Intermediates of levosulpiride.
Niksan Pharmaceutical provides best quality product of levosulpiridein all states of India like Kerala, Punjab, Rajasthan, Telangana, Bihar,Karnataka, Jammu and Kashmir, Maharashtra, West Bengal, Uttar Pradesh, Gujarat,Delhi, Tamil Nadu, Odisha, Madhya Pradesh, Andhra Pradesh, Haryana and many other states.
Niksan Pharmaceutical exporting very big quantity of the fines quality products of levosulpiride in all over world formany years in countries like Afghanistan,Paraguay, Nicaragua, Honduras, Pakistan, Costa Rica, Panama, Dominican Republic, Venezuela, Cambodia, Guatemala, Italy, Vietnam, Peru, South Korea, Bolivia, United Arab Emirates, Colombia, Ecuador, Chile, Spain, Mexico, United Kingdom, United States, Germany and many more countries.
Levosulpiride is antipsychotic agent which is used in the mood elevating treatmentslike mood swings, depression. Levosulpiride also used in the treatment toimprove the food movements and gut movements.
IUPAC NAME: N-[[(2S)-1-ethylpyrrolidin-2-yl]methyl]-2-methoxy-5-sulfamoy
CAS NO: 23672-07-3
FORMULA: C15H23N3O4S
MOLECULAR MASS: 341.4g/mol
STORAGE CONDITION: Store in cool and dry place, away from light anddirect heat. Do not place medicine in bathroom or humid place. Keep away fromchildren and pets
Â
HOW TO USE: Take this medicine before meal one time aday. Take this medication in morning without food. Do not consume alcohol whiletaking the medication.
Â
HOW LEVOSULPIRIDE WORKS: Levosulpiride increase the release ofacetylcholine in body.by this process acetylcholine increase the movement ofstomach and intestine and prevents the reflux. Levosulpiride also improvesstomach pain, abdominal cramps and intestinal problems.
Â
PHARMACOKINETICS: Levosulpiride absorbed rapidly in bodyafter the oral administration. It takes 30 min to reach at peak plasmaconcentration. Only 35% of drug binds with the plasma proteins. Levosulpiridebinds with the dopamine receptors and gives effects. The half-life of thismedicine is 7 hours. The elimination of Levosulpiride is done by renal route.
Â
SIDE EFFECTS: The side effects of Levosulpiride aredrowsiness, constipation, weight gain, insomnia, fever, nausea, vomiting, andheadache. Contact your doctor if you see side effects like abdominal pain,increased salivation, Gynacomasti, irregular menstrual periods, breasttenderness, change of heart rate, weakness.
Â
PRECAUTIONS: Before taking the medication tell yourdoctor if you are allergic to the medicine. Tell your medical history if youhave diabetes, heart problem, liver problem, abdominal abnormalities, andbreast cancer. Do not take medication if you are pregnant or in lactationperiod. Avoid the medication if you are breast feeding to new born baby.
Do not take alcoholor cannabis while taking the medicine.
Â
CDSCO APPROVAL: Levosulpiride Tablet (50mg/100mg) (addl. strength for addl.indication) approved by CDSCO in India in 12.6.2007
 Levosulpiride Inj. 25mg/ml,2ml approved by CDSCO in India in26.09.2008
 Levosulpiride 75/150/200 mg(add. Strength)approved by CDSCO in India in 09.12.2014
Levosulpiride tablet (25mg) approved by CDSCO in India in10.05.2007
Â
FORMULATIONS AVAILABLE IN MARKET:
Levosulpiride 50 mg tablets
Levosulpiride 100mg tablets
Levosulpiride 25mg/ml injection
Levosulpiride 2ml injections
Levosulpiride 75mg tablets (add. Strength)
Levosulpiride150mg tablets (add. Strength)
Levosulpiride 200mg tablets (add. Strength)
Levosulpiride75mg + Rabeprazole 20mg tablets
Levosulpiride 75mg + pantoprazole40mg tablet s
Levosulpiride 12.5mg/ml injections
Levosulpiride 75mg + Pantoprazole 80mg tablets
Levosulpiride25mg + Rabeprazole 20mg tablets
Levosulpiride75mg + Rabeprazole 40mg tablets
Â
Note: Product protected by valid patents are notoffered for sale in countries where such patents are still valid and itsliability is at Buyers Risk
Â
REFERENCES:
Â
www.webmd.com
https://pubchem.ncbi.nlm.nih.gov
https://go.drugbank.com
https://cdscoonline.gov.in
https://www.wikipedia.org/
https://www.drugs.com
https://www.zaubacorp.com
https://www.practo.com/consult

Price:
- 50
- 100
- 200
- 250
- 500
- 1000+
Other Products in 'Active Pharmaceuticals Ingredients (API)' category
 |
NIKSAN PHARMACEUTICAL
All Rights Reserved.(Terms of Use) Developed and Managed by Infocom Network Private Limited. |
 English
English Spanish
Spanish French
French German
German Italian
Italian Chinese (Simplified)
Chinese (Simplified) Japanese
Japanese Korean
Korean Arabic
Arabic Portuguese
Portuguese